|
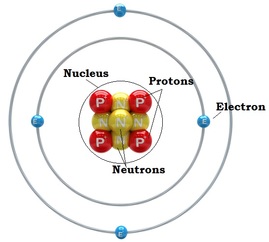
Today in class, Andrew gave us an opportunity to get points. He did an activity on Chemical Reactions, which later became a class wide assignment. Purpose: Discovering more about chemical reactions and the periodic table of elements, also making H20 with Combustion Group Members: Griffin, Israel, Juan ____________________________________________________________________________________________________ Beforehand Lesson: Andrew briefed us on chemical reactions and the periodic table before conducting the actual experiment in our groups. What is a chemical reaction?
What is a combustion reaction?
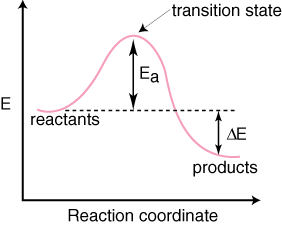
Thermodynamics of Chemical Reactions
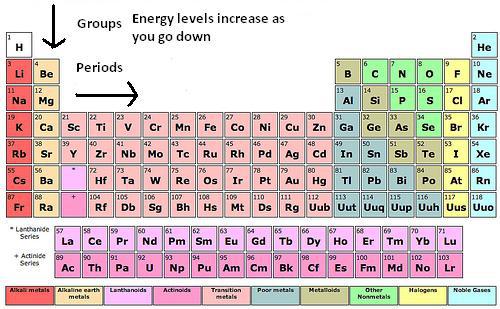
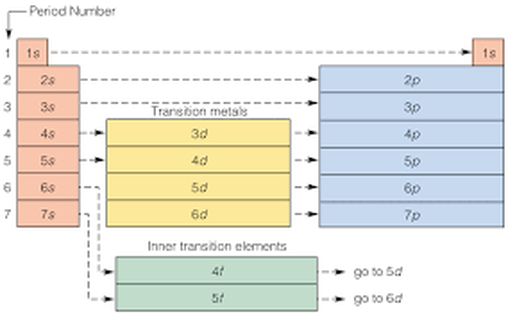
Periodic Table of Elements:
Materials:
Directions:
What happened? A Chemical Reaction Story So I did this project assignment with Griffin, Israel and Juan. After we acquired our necessary materials. We filled up the beaker with water and began to mix in the citric acid. Of course nothing happened, but then Andrew passed around the coil of magnesium. He demonstrated how to coil the long strands and then Griffin followed and coiled it for us. We dropped it in. Slowly it appeared as if it were boiling in the beaker. The liquid itself was bubbling and when we all tried placing our hands on it, we felt that the beaker itself was gradually getting warmer. After awhile we noticed a strong scent in the air. We realized it was from the magnesium in the citric acid. We cringed at the burning scent for it was not exactly a pleasant one. Andrew then passed out the black balloons. We put over the balloon onto the beaker. We waited for the balloon to slowly fill up with the hydrogen exerting from the chemical reaction between the magnesium and the citric acid. Slowly the balloon filled up, not as big as a standard sized party balloon, but to about a circumference of 3 1/2 inches. When we said the balloon was filled up, Andrew tied it off and let us play with the balloon. The balloon was floating on its own and if we weren't careful, it would fly up towards the ceiling like another person's did. By the time we were done playing with it, we went to go light up the balloon to see the combustion reaction happen. One by one we waited in line to light up our table's balloons. When it was our turn, Israel tied the balloon to a long beam that he would later place next to the fire to make it explode. Andrew turned of the lights and with a match, lit up a birthday candle on the table. Israel slowly dropped our balloon onto the match, and at the very second it made contact with the fire, there was a mini explosion. THE COMBUSTION REACTION was a success. There was hydrogen in the balloon due to the chemical reaction between magnesium and the water/citric acid. And we filled up the balloon with the hydrogen energy. Then we lit up the balloon so it could exert the energy out into a small scale explosion. The energy radiating from the match was the enough amount of energy (aka the activation energy) to initiate the chemical reaction. What I learned? Noticed?
Afterthoughts: I thought the whole experiment went well. At first I couldn't understand the examples he was givingon the periodic table of elements. But afterwards I understood it. The actual things we learned were really helpful, I learned a lot about periodic table which will be very helpful for the future. I thought that surprisingly our balloon filled up the most. It was the biggest balloon, why is that though? I thought overall we did well, nothing could have really gone wrong. It was cool seeing the balloon float on its own. It's hard to explain how everything works in general but it's slowly becoming easier to understand after doing more research and more worksheets on it. It was cool seeing the explosion happen due to the combustion reaction. It kind of made me think about how much more of an explosion we could make if we made a reaction on a much greater scale. What would you do differently? I would probably have time lapsed the burning sensation of the magnesium to the citric acid. I would have documented a lot more, regarding the notes we took on the research beforehand and also the whole experiment as a whole. Next time I want to document more and I want to not procrastinate when it comes to blogging.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2015
Categories |


 RSS Feed
RSS Feed
