|
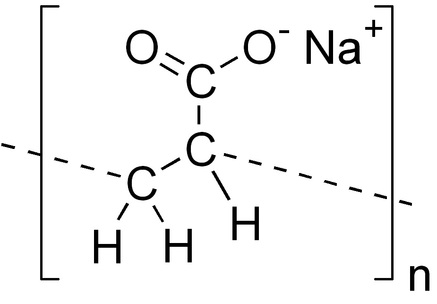
Purpose: To experiment on Sodium Polyacrylate and to try to expand the mass of it by adding water, creating "Fake Snow". Group Members: In this project I worked with Sammie. ____________________________________________________________________________________________________ Background Research: Sodium Polyacry is?
Pre-Lab/Safety Precautions: Potential Health Hazards
Materials:
Directions:
What happened? A fake snow story... Sammie and I decided to work together on this project again. We first put on safety glasses, then Braden corrected us and made us wear masks. Embarrassed, we pulled the masks on tight and began our work. We got one diaper, and like we were told, we opened up the diaper to acquire our sodium polyacrylate. We then began to extract the cotton, as you can see on the bottom right, we teared off the cotton, leaving the sodium polyacrylate to be extracted into our beaker. Then we got our sodium polyacrylate and scraped it into the beaker. It took awhile to get a good amount of the dust. We had to basically tear our diaper apart to get all we could. It was often scattered around the diaper. We poured a good amount of it into our beaker, we knew that we didn't need sooo much since the sodium polycrylate itself expands much more than in its current state. After getting a small amount of sodium polycrylate in our beaker, we then poured it into a bowl to start mixing it with H20. It slowly absorbed it when we began mixing it with our fingers. They popped up like little ball sponges. This time lapse shows our mixing process. Already we saw a big difference in the sodium polycrylate. It started off just barely filling up a layer of dust in our foam bowl. And already, after adding water, it filled up the whole bowl. We first got a consistency like the one below. A "snow" like consistency. Where it is more of the solid state of matter. We then thought about what to do next. Sammie suggested that we add more water. I filled up a small beaker with water and threw it in with the "snow" like sodium polyacrylate. We wanted to just add in little by little, but later, after adding 400 ml of water, we had to move our snow to a much larger container. It began to leak and overflow out of the bowl, and so we got a large glass container. So at this point, the consistency is very "slushy" like. It appears like icy water. And at this point the state it is in is more of the liquid side, it can't hold a certain shape or volume. Like the water, the temperature of the actual sodium polyacrylate was cold. We finished our experiment at this point.
___________________________________________________________________________________________________ What I learned/noticed?
Questions I had/researched?
If I could do it differently?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2015
Categories |






 RSS Feed
RSS Feed
