|
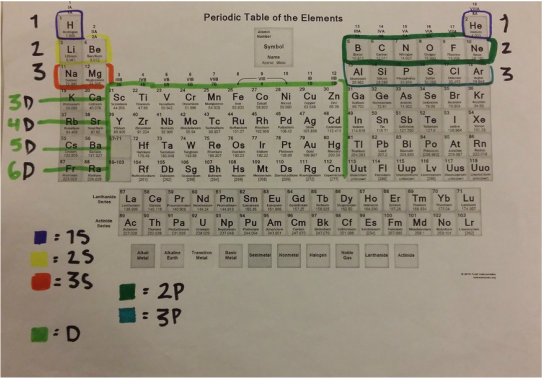
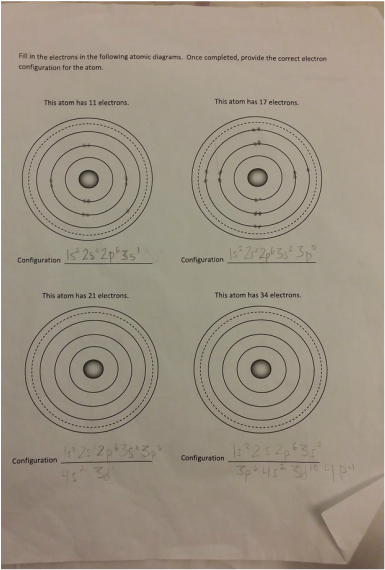
Overview: I really felt like I learned a lot from this work sheet. It helped because we recently did an activity related to the periodic table of elements. I was very confused of how to do the second part of this worksheet at first. I understood the part's of the atoms but after the first page I didn't know how to place the electrons on the atom. After asking Braden he totally enlightened me on how to do it. I was so thankful that he explained it to me, now I understand it really well and it is very good for me to know this for the future. THANK YOU SO MUCH BRADEN! The Periodic Table: Here is the cheat sheet I made of the periodic table of elements. It has different colors showing each "shell" of the elements. To label the configuration I learned that you have to state the Shell Letter which is either S, P, or D, and also how many electrons in the shell. Let's say there is both electrons for the second shell, you would label a exponent 2 for 2P. I learned that on the atom diagram, there are 8 shells and that each of them represent the rows of the periodic table of elements. There is an S side of the periodic table that is on the left. It has Hydrogen and Helium for the first shell (the only exception for S to have electrons from both the left and right of the table), and the rest are going down in pairs of 2. There is a P side of the periodic table on the right, in which Helium is not a part of. It goes in 6' s down for the P. There is also a middle part of the periodic table, it starts on the the 3rd column up until the 12th column. It is the D side of the periodic table. Braden told me that instead of lining up with S and P and starting at 3, it starts at 4D. While S is 3S, D IS 4D. Worksheet: For example: CHLORINE How to get it: The Answer: 1s2, 2s2, 2p6, 3s2, 3p6
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2015
Categories |

 RSS Feed
RSS Feed
